Пірыдаксал гідрахларыд (65-22-5) Sикация
| імя: | пиридоксаль гідрахларыд |
| CAS: | 65-22-5 |
| Чысціня | 98% |
| Малекулярная формула: | C8H10ClNO3 |
| Малекулярная вага: | X |
| Падпаліць Point: | 173 ° C |
| Хімічнае назва: | ГІДРАХЛАРЫД ПІРЫДОКСАЛУ Пірыдаксаль HCl 3-гідраксі-5- (гідраксімэтыл) -2-метилизоникотинальдегид гідрахларыд |
| Сінонімы: | 3-гидрокси-5- (гидроксиметил) -2-метил-4-пиридинкарбоксальдегид гідрахларыд / пірыдаксал HCl |
| InChI Key: | FCHXJFJNDJXENQ-UHFFFAOYSA-N |
| Палова жыцця: | N / A |
| растваральнасць: | Раствараецца ў вадзе |
| Умова захоўвання: | 0 - 4 C на кароткі тэрмін (ад дзён да тыдняў), альбо -20 C на працяглы тэрмін (месяцы) |
| Ужыванне: | Гэта неабходныя для падтрымання здароўя нерваў, скуры і эрытрацытаў. Пірыдаксін выкарыстоўваўся для прафілактыкі або лячэння пэўнага нервовага расстройства (перыферычнай нейропатии), выкліканага некаторымі лекамі (напрыклад, изониазидом). |
| Знешні выгляд: | Парашок белага колеру |
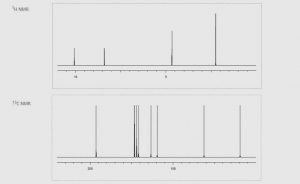
Пірыдаксал гідрахларыд (65-22-5) Спектр ЯМР
Калі вам патрэбна COA, MSDS, HNMR для кожнай партыі прадукту і іншая інфармацыя, звяжыцеся з намі менеджэр па маркетынгу.
Што такое пірыдаксаль гідрахларыд?
Пірыдаксаль гідрахларыд - гэта форма вітаміна В6. Як 4-карбальдэгідная форма вітаміна В6, ён вырабляецца шляхам змешвання пірыдаксалю з адным малярным эквівалентам салянай кіслаты. Гэта харчовая дабаўка. Яго хімічная формула - C8H10ClNO3. Яго назва IUPAC-3-гідраксі-5- (гідраксіметыл) -2-метылпірыдын-4-карбальдэгід гідрахларыд.
Гэта рэчыва адносіцца да класа арганічных злучэнняў пад назвай пірыдаксаль і яго вытворных. Яны ўтрымліваюць пірыдаксальны фрагмент з пірыдынавым кольцам, якое мае замену ў розных кропках. Гэтыя кропкі замяшчэння ўключаюць метыльную групу ў пазіцыі 2, гідраксільную групу ў пазіцыі 3, карбальдэгідную групу ў пазіцыі 4 і гідраксіметыльную групу ў пазіцыі 5.
Пірыдаксаль гідрахларыд выкарыстоўваецца пры лячэнні дыябетычнай нефрапатыі. Гэта неабходна для падтрымання здароўя нерваў, скуры і эрытрацытаў. Ён выкарыстоўваецца арганізмам для вытворчасці нейрамедыятараў, такіх як серотонін і норадреналін. Гэта таксама дапамагае выпрацоўцы сфінгаліпідаў і аміналевулінавай кіслаты. Пірыдаксаль гідрахларыд ператвараецца ў 5-фасфат пірыдаксалю, кафермент для сінтэзу амінакіслот.
Так як вітамін B6 не можа выпрацоўвацца ў арганізме трэба спажываць з розных крыніц або праз дабаўкі. Ён існуе ў некалькіх формах, адной з іх з'яўляецца пірыдаксаль, які з'яўляецца 4-карбоксальдэгіднай формай вітаміна В6, а таксама кофактором для шматлікіх метабалічных дзеянняў.
Пірыдаксаль гідрахларыд - гэта солевая форма пірыдаксалю і лёгка ператвараецца ў пірыдаксальфасфат, які пасля ўвядзення таксама вядомы як ПЛП, і функцыянуе як кафермент для метабалічнай актыўнасці.
Як дзейнічае пірыдаксаль гідрахларыд?
Вітамін В6 у прыродзе мае тры формы: пірыдаксін, пірыдаксаль і пірыдаксамін, усе яны ператвараюцца ў актыўную форму ў арганізме і называюцца 5'-фасфатам пірыдаксалю. Сімптомамі дэфіцыту вітаміна В6 з'яўляюцца себорейный дэрматыт, микроцитарная анемія, глоссит, курчы, перыферычная нейропатия, дэпрэсія і г. д. Вітамін В6 таксама прызначаецца для лячэння перадазіроўкі изониазида, атручвання ілжывымі марскімі грыбамі, ўздзеяння гідразіну і г. д. Пірыдаксаль гідрахларыд можа дзейнічаць як дадатак для папаўнення ў арганізме вітаміна В6.
Пірыдаксаль гідрахларыд- папярэднік 5'-фасфату пірыдаксалю. Пасля спажывання пірыдаксаль гідрахларыд ён ператвараецца ў 5-фасфат пірыдаксалю ў гепатацытах і слізістых клетках кішачніка. Затым ён паглынаецца крывацёкам і размяркоўваецца па ўсім целе. Ён удзельнічае ў мностве метабалічных рэакцый. Гэта ўключае адукацыю і абмен амінакіслот і глікагену. Гэта таксама можа дапамагчы ў сінтэзе нуклеінавых кіслот, гемаглабіну і нейрамедыятараў.
Пірыдаксаль 5'-фасфат-гэта кафермент, які ўдзельнічае ў абмене амінакіслот. Ён паводзіць сябе як кафермент ва ўсіх рэакцыях трансамінавання. Ён таксама ўдзельнічае ў рэакцыі акіслення і дэзамінавання амінакіслот.
Альдэгідная група, прысутная ў 5'-фасфаце пірыдаксалю, утварае сувязь на аснове Шыфа з эпсілон-амінагрупай спецыфічнай групы лізінаў ферментаў амінатрансферазы. Затым альфа-амінагрупа амінакіслотнага субстрата выцясняе эпсілон-амінагрупу. Гэта прыводзіць да ўтварэння адыміну, які дэпратонуецца. Пасля гэтага ён становіцца хиноидным прамежкавым прадуктам, які прымае пратоны ў розных пазіцыях і, нарэшце, становіцца кетимином. Затым кетымін падвяргаецца гідролізу, так што амінагрупа застаецца ў бялковым комплексе.
Пірыдаксаль гідрахларыд таксама дапамагае сінтэзаваць нейрамедыятары, такія як серотонін, дофамін, норадреналін і гама-амінамасляная кіслата.
Пірыдаксаль гідрахларыд яшчэ не быў ухвалены FDA.
сінтэз
Пірыдаксаль гідрахларыд сінтэзуецца з арганічнай рэакцыі шляхам рэакцыі пірыдаксаміну з адным малярам салянай кіслаты. Пірыдаксаль гідрахларыд можна таксама сінтэзаваць шляхам выбарачнага акіслення. Рэакцыя складаецца з узяцця пірыдаксіну гідрахларыду ў якасці зыходнага матэрыялу, і ў вадзе будзе праведзены метад каталітычнага акіслення. Каталітычнае акісленне ўключае крыніцу кіслароду, каталізатар, неарганічную соль і амінавы ліганд. Канчатковым прадуктам каталітычнага акіслення быў бы гідрахларыд пірыдаксалю.
Фармакакінетыка пірыдаксаль гідрахларыду
паглынанне: Яны лёгка ўсмоктваюцца пры пасіўнай дыфузіі, і іх паглынанне ў асноўным лакалізуецца ў клетках слізістай абалонкі кішачніка, багатых пірыдаксінкіназай і пірыдаксальфасфатам.
Метабалізм: Большасць з гэтых арганічных злучэнняў паглынаецца печанню, і выніковае паглынанне ажыццяўляецца за кошт носьбітаў-апасродкаванай дыфузіі і метабалічнага захопу ў выглядзе фасфатных злучэнняў. Фасфараляванне злучэнняў лёгка адбываецца ў печані.
Распаўсюд: Свабодны пірыдаксаль фасфат у печані гідралізуецца да пірыдаксалю, які пазней экспартуецца і звязваецца з гемаглабінам і альбумінам у эрытрацытах. Дефосфорилированная частка пакідае клетку шляхам дыфузіі, і, такім чынам, у тканінах будзе мала назапашвацца пірыдаксаль фасфат.
Выдаленне: Вольны пірыдаксаль, які застаецца ў печані, хутка акісляецца да 4-пірыдаксавай кіслаты і з'яўляецца асноўным прадуктам вылучэння злучэння. Акісленне да 4-пірыдаксінавай кіслаты ажыццяўляецца з дапамогай альдэгіддэгідрагеназы, якая шырока прысутнічае ў шматлікіх тканінах, акісленне таксама ажыццяўляецца ферментамі альдэгідыдаксідазы нырак і печані. Ключавы шлях вывядзення пірыдаксаль гідрахларыду пасля метабалізму - праз мачу.
Перавагі пірыдаксаль гідрахларыду
Ёсць некалькі відаў прымянення пірыдаксаль гідрахларыду. Аднак большасць з гэтых ужыванняў дагэтуль даследуецца і не можа лічыцца пэўным ужываннем гэтага злучэння.
Вось некаторыя з пераваг пірыдаксаль гідрахларыду
Ўплыў на дыябетычную нефрапатыю
Атрыманы прадукт пірыдаксаль гідрахларыду пірыдаксаль 5'-фасфат можа інгібіраваць адукацыю канчатковых прадуктаў пашыранага гликирования (ВУР). Ён робіць гэта, захопліваючы 3-дэзаксіглюказон. У даследаванні з дыябетычнымі пацукамі, індукаванымі стрэптазотацынам, іх лячылі пірыдаксаль-5-фасфатам на працягу 16 тыдняў [1]. Вынікі паказалі значнае зніжэнне альбумінурыі, гіперметропіі клубочкаў, мезангиальное пашырэнне і міжтканкавы фіброз. Гэта таксама знізіла ўзровень узросту. Такім чынам, у доўгатэрміновай перспектыве пірыдаксаль гідрахларыд можа знізіць нефрапатыю, якая ўзнікае пры дыябеце.
Эфект як метабаліт
Пірыдаксаль гідрахларыд і пірымідын 5'-фасфат дзейнічаюць як жыццёва важныя рэчывы, неабходныя падчас метабалічных рэакцый. Іх здольнасць дзейнічаць у якасці каферменту патрабуецца абодвум чалавечага цела а таксама розныя арганізмы і бактэрыі, такія як кішачная палачка, Saccharomyces cerevisiae, мышы. Яны неабходныя для выпрацоўкі розных злучэнняў, неабходных арганізму.
Ўздзеянне на нейрамедыятараў
Пірыдаксаль гідрахларыд можа дапамагчы ў стварэнні розных нейрамедыятараў ў арганізме.
Ўплыў на анемію
Пры некаторых анеміях вітамін В6 неабходна забяспечыць у якасці дадатку. Пірыдаксаль гідрахларыд можа дапамагчы забяспечыць неабходны 5'-фасфат пірыдаксалю для лячэння такіх станаў.
Выкарыстанне ў медыцынскіх даследаваннях
Пірыдаксаль 5'-фасфат, атрыманае злучэнне пірыдаксаль гідрахларыду, неабходны для некаторых бактэрый, якія маюць значэнне з медыцынскай пункту гледжання. Гэта для забеспячэння іх правільнага росту. Яна ўключае ў сябе такія бактэрыі, як гранулікатэла і абіятрофія [2]. Харчовая патрэба пірыдаксаль гідрахларыду можа выклікаць культурны феномен росту спадарожнікаў у гэтых бактэрый. У культуры in vitro гэтыя бактэрыі могуць расці толькі ў тых месцах, дзе ёсць іншыя бактэрыі, якія ўтвараюць пірыдаксаль. Адзін з галоўных момантаў злучэння пірыдаксалю заключаецца ў тым, што яно, магчыма, ператварылася ў самую старажытную рэакцыю аэробнага метабалізму на зямлі.
Пабочныя эфекты пірыдаксаль гідрахларыду
- раздражненне скуры
- падвышаная адчувальнасць
- Раздражненне дыхальных шляхоў
- Млоснасць
- Ваніты
- Перыферычная нейропатия
- Зніжэнне адчувальнасці
- Галаўны боль
- неадчувальнасць
- паколванне
- нястраўнасць
Узаемадзеянне пірыдаксаль гідрахларыду з іншымі лекавымі сродкамі
Інфармацыі аб прамым узаемадзеянні пірыдаксаль гідрахларыду з іншымі прэпаратамі недастаткова. Аднак вядомая інфармацыя аб узаемадзеянні яго атрыманага злучэння пірыдаксаль 5'-фасфату з іншымі лекамі.
Некаторыя з гэтых узаемадзеянняў:
Аміёдарон -Пры ўзаемадзеянні з 5'-фасфатам пірыдаксіну ён можа павялічваць адчувальнасць да святла, выклікаючы сонечныя апёкі і г.д.
Фенитоин -Пірыдаксін 5'-фасфат можа павялічваць метабалізм фенітаіну ў арганізме, выклікаючы меншую эфектыўнасць апошняга.
фенабарбітал -Пірыдаксін 5'-фасфат можа павялічыць час распаду фенабарбіталу.
леводопа - Гэта можа выклікаць хуткі метабалізм леводопы.
У той час як вядома, што дэфіцыт пірыдаксін ўзнікае падчас цяжарнасці і груднога гадавання, гэта не так інфармацыя аб эфектах пірыдаксаль гідрахларыд у гэтых умовах. Таму рэкамендуецца праяўляць асцярожнасць.
Дзе купіць пірыдаксаль гідрахларыд у 2021 годзе?
Вы можаце купіць парашок гідрахларыду пірыдаксалю непасрэдна ў кампаніі -вытворцы пірыдаксаль гідрахларыду. Ён даступны ў колькасці 1 кг за мяшок або 15 кг за барабан. Аднак гэта можна наладзіць у адпаведнасці з патрэбамі карыстальніка. Гэты парашок неабходна захоўваць пры тэмпературы ад 0 да 4 ° на кароткі тэрмін і пры -20 ° С на працяглы тэрмін.
Парашок пірыдаксаль гідрахларыд выпускаецца ў выглядзе парашка белага або брудна-белага колеру. Ён зроблены з лепшых інгрэдыентаў пад строгім наглядам, каб гарантаваць, што карыстальнікі атрымаюць самы лепшы прадукт.
Спасылкі
- Nakamura, S., Li, H., Adijiang, A., Pischetsrieder, M., & Niwa, T. (2007). Пірыдаксаль фасфат прадухіляе прагрэсаванне дыябетычнай нефрапатыі. Нефралогія Дыяліз Трансплантацыя, 22(8), 2165-2174.
- Кітада, К., Окада, Ю., Канамота Т., Інуэ, М. (2000). Сералагічныя ўласцівасці відаў Abiotrophia і Granulicatella (варыянт страўнікавага стрэптакока). Мікрабіялогія і імуналогія, 44(12), 981-985.






